Mucosal vaccines go directly into the mucosal tissue where infection begins – for example, intranasally or via tablets. If they could induce…
First in Human: Covid-19 Vaccines & Tales of Phase 1 Clinical Trials Past

The people who sign up for the first-in-human trials of Covid-19 vaccines are about to do our societies an extraordinary service. Most of the people who could do it, would be at best reluctant to – even among those who are willing to participate in other kinds of first-in-human trials (Chen 2017). Here are a couple of them talking about why they are doing it:
They are part of a phase 1 clinical trial for a COVID-19 vaccine aiming for originally for 45 participants, that was registered and recruiting on 25 February 2020, with the first dose injected on 16 March: on 19 March they had all the people they needed, and closed to recruitment. The NIH Director reported that the vaccine has been tested in animals. The first healthy volunteers come from Seattle in the US – a giant thanks to all of them! On 20 March, a second site for the trial was added the trial’s registry entry: a pediatric hospital in Decatur, Georgia. On 30 March, the registry entry noted the Georgia site was now recruiting, and a third location was added at the NIH’s Clinical Center in Bethesda (not yet recruiting).
This vaccine and trial have gotten going incredibly fast for a disease that only emerged late last year. The trial’s registration entry reports it will test the safety and immune response of various doses of an mRNA-based vaccine (currently called mRNA-1273), following people for a year after the second of the 2 injections they will get. (More details in the NIH Director’s report.)
The trial is sponsored by the NIH’s National Institute of Allergy and Infectious Diseases (NIAID). If it makes it through to FDA approval, it would be the first mRNA-based vaccine in use, using genetics to instruct cells to produce antibodies. Being the first in the US means it should face extra scrutiny if it is to get that far. On 7 May, the company announced it had FDA approval to go onto a Phase 2 trial, and it’s aiming to start a Phase 3 trial “early this summer”. The clinical trial register entry for the trial was updated, increasing the number of participants in the Phase 1 trial to 105, and pushing back the completion date.
There are other types of vaccine in preclinical development too. For more about Covid-19 vaccines, see Laura Spinney’s article. And for how things are going, check out my monthly roundup posts.
On 18 March, a trial of a different recombinant Covid-19 vaccine was registered. That vaccine is based on adenovirus Type 5 Vector (currently called Ad5-nCoV), and is recruiting 108 adults in China. It’s following up for 6 months, so could have some results before the NIH’s Seattle/Decatur/Bethesda trial. A Phase 2 randomized trial beginning 12 April was subsequently registered for this vaccine. That trial is aiming to enrol 500 participants, in 3 arms: 250 receiving a middle dose, 125 a low dose, and 125 a placebo injection, with 6 months follow-up. There was also a high dose group in the Phase 1 trial. No results have been reported: the manufacturer’s report to the Hong Kong stock exchange says it is proceeding “based on the preliminary safety data of the phase 1 clinical trial”.
Scientists from Oxford University are fast-tracking another adenovirus vector vaccine, called ChAdOx1 nCoV-19 (more details here). They are leap-frogging straight to a Phase 1/Phase 2 trial and hope to beat the 12-18 months estimate till a vaccine is available. On 27 March, they announced they are recruiting participants 510 healthy people, with 260 to be randomized to the vaccine and the other 250 to a saline injection. It will be a 6-month trial, and although they are already recruiting, preclinical animal testing is still going on. If that goes well, then the first-in-human randomized trial will begin: their timetable for that is a few weeks. (Clinical trial register entry here. In a 14 April update, they reported they will be recruiting in Southampton and London as well as Oxford. On 23 April, the first injections in this trial were reported.)
And a fourth. According to the company manufacturing it, the first dose of this trial was given to a healthy volunteer on 6 April 2020. Currently called INO-4800, this one is partly funded by the Gates Foundation, and it builds on previous work on a vaccine for another coronavirus, MERS. This Phase 1 trial aims to recruit 40 people, at 2 sites in the US: Philadelphia and Kansas City, with 6 months follow-up (to November 2020). The clinical trial register entry doesn’t tell you anything about the vaccine itself, but unlike the others, there is a preprint about its development and initial animal tests. It’s a DNA vaccine, delivered through a hand-held device that’s geared to be feasible in at-risk countries globally: trials in China and South Korea are also planned.
The fifth was the first German trial. It received approval to start a Phase 1/Phase 2 trial on 20 April. This is a 2-part dose-escalation trial with parallel cohorts in 196 people, testing 4 RNA vaccines called BNT162 a1, b1, b2, and c2. It will be run in multiple sites in Germany, and with further studies are planned in the USA and China. (Clinical trial register entries: for Germany on 24 April; for USA on 5 May.) The first dose in the US was announced on 5 May.
Also on 20 April, another Phase 1/Phase 2 trial was registered. This inactivated SARS-CoV-2 vaccine is unnamed, and is being run in Xuzhou, China (Jiangsu province). The Phase 1 trial aims to recruit 144 people, and the Phase 2 trial aims for 600, randomized to the candidate vaccine or placebo. (A preprint on this vaccine’s preclinical phase was posted on 19 April.)
The first Canadian vaccine trial is planned to get underway at 2 universities in British Columbia on 30 April. This is for an oral vaccine, named bacTRL-Spike, which uses SARS-CoV-2 DNA on a bacterial medium. It’s a Phase 1 trial with 84 planned participants, randomized to either the vaccine or the bacterial medium alone. (Clinical trial register entry.)
On 28 April, there was a call for participants in Perth, Australia, for a Phase 1 trial of a vaccine called Covid-19 S-Trimer. This vaccine uses antibodies from people who have recovered from Covid-19.
A Phase 1 trial is scheduled to start on 15 May in Brisbane, Australia, for a nanoparticle vaccine called SARS-CoV-2 rS. (Clinical trial register entry.) It will aim for 131 people, testing the vaccine with and without a trademarked adjuvant, versus a placebo (saline) injection. It might test a second vaccine, too.
First-in-human vaccine trials
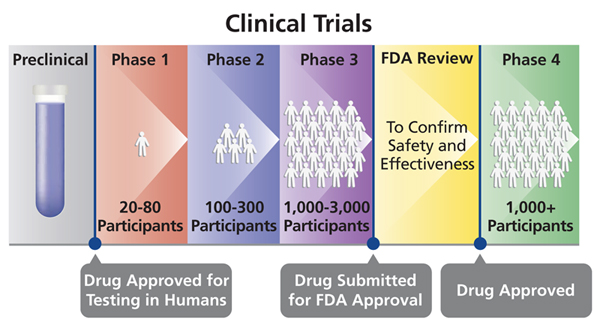
Let’s go back to the beginning. The preclinical phase involves all the work that goes on in laboratories to develop a vaccine. A phase 1 clinical trial is a small, first-in-human test of a drug or device, testing safety and response in carefully selected people who are believed to be at relatively low risk of being harmed. For example, the Seattle trial’s registry entry lists 15 criteria you have to meet to be included, and 25 reasons you could be excluded even if you meet them.
A phase 1 trial can show if a vaccine could work, but it’s just a start: it’s not enough to test its benefits or harms in the general community. The graph below is from the NIH. It shows where these trials fit into the process in the US, but the process is similar in other parts of the world. You need phase 3 trials to get a vaccine or other drug approved.
These days, phase 1 trials are often done by commercial organizations. The healthy people who volunteer for them are far more likely to be on a low income or unemployed than the general population. That’s according to a study of 1,194 people in one company’s drug trials in Belgium, Singapore, and the US (Grady 2017), and Chen’s study points to other signs of this as well. That means financial pressure might be pressing people into doing it: they might really need the payment they get for participating in these trials.
Ethicist Carl Elliott wrote an article in The New Yorker called “Guinea-Pigging” in 2007, saying that trials in the private sector have “created a new dynamic. The relationship between testers and test subjects has become, more nakedly than ever, a business transaction”.
That isn’t the case for this first Covid-19 vaccine trial. It is being run by the non-commercial Kaiser Permanente Washington Health Research Institute, open to people living or working in Seattle. Participants in the trial will reportedly receive $100 for each of up to 11 visits. The people who step up will be joining a long history of people shouldering a serious load for the rest of us. Let’s take a quick look at some tales of first-in-human vaccine tests – starting with the most famous and mythologized case, smallpox. Most of the popularized story about it is wrong.
Here’s the legend that grew around this: Jenner got the idea for using cowpox to vaccinate because milkmaids had beautiful skin untouched by smallpox. He picked out his gardener’s son to be the first person experimented on with this hypothesis, and then infected him with smallpox to see if it worked. None of that is true. And the real story is far more interesting.
Before Jenner in 1796, Benjamin Jesty may have been first in England in 1774, then Peter Platt in Germany in 1791. Back then, people inoculated children with actual smallpox – that had happened to Jenner himself when he was 8. Arthur Boyle has written the story of what really happened with Jenner and Phipps on the James Lind Library. It wasn’t a single “Eureka!” moment. Others in his professional circle had already considered and more or less dismissed the cowpox hypothesis. Jenner stuck with it, though. Boyle writes:
It took Edward Jenner twenty-five years to unravel the mess. The problem lay in the definition of ‘cowpox’. There were at least three diseases that produced ulcers on the teats of cows and only one of these was caused by the cowpox virus: ‘this for a while damped, but did not extinguish my ardour’. Jenner’s remarkable achievement is that he spent all that time untangling true cowpox from spurious cowpox and defining its unequivocal appearances. He learned how to recognise ‘milker’s nodes’ a painful bacterial infection on the fingers, which lacked the typical erosive ulcer of cowpox, and he could differentiate between cowpox and staphylococcal infections of the udder because the damage caused by staphylococcal bacteria spread beyond the teats. Eventually he learned how to recognise cases of true cowpox and could confirm that they really were resistant to inoculation. Jenner had also had a second insight that his colleagues had not appreciated. Inoculated cowpox would be much less severe than natural cowpox, just as inoculated smallpox was less dangerous than naturally acquired disease.
He didn’t seek James Phipps out: Phipps’ father asked Jenner to inoculate him. Boyle again:
Although Jenner had never ‘vaccinated’ anyone before he had good reason to believe that the procedure might work based on numerous patients of his who had proved resistant to inoculation after having cowpox. All he was doing was attempting to see whether artificial cowpox worked as well as natural cowpox, just as artificial inoculated smallpox protected against natural smallpox. After vaccinating Phipps he was be inoculated in the usual way. If cowpox had failed, he would just have a normal mild inoculation response; if it worked nothing would happen. Jenner was following up his experimental treatment with the ‘gold standard’ method of protecting the child. Since inoculation was a well-established and largely safe procedure that was widely used in England, there was no ethical issue with that part of the experiment. Inoculation would have been viewed as a necessary part of growing up for a child receiving parish support. Since cowpox was never fatal and had few systemic effects, there were unlikely to be any unexpected complications apart from failure to immunize. Even in the hyper health-and-safety conscious twenty-first century this experiment would have been given ethical approval.
Eventually, Jenner provided the adult Phipps and his family with a lease-free cottage, and it’s a museum now. Here’s a plaque on its wall (photo by Gaius Cornelius):
James Phipps may not have been part of Jenner’s household, but someone experimenting on a family member or a member of their household community was often how first-in-human vaccines were tested. Thomas Jefferson later tested his smallpox vaccine on enslaved people, for example, going on to vaccinate family and neighbors.
But another common first human is the scientist who developed the vaccine. Let’s fast-forward about 100 years to one of them, Waldemar Haffkine. He was born to an educated Jewish family in what’s now Ukraine, and named Vladimir Aronovich Mordecai Wolf Chavkin. As a university student, he became politically active during pogroms, and was wounded and imprisoned. Eventually, he had to leave what was then the Russian Empire, arriving at the Pasteur Institute in Paris after a stay in Geneva. His job was in the medical library, but in his spare time he worked on a vaccine for cholera.
Haffkine found that if he passed the cholera bacilli 39 times through guinea pigs, he had a culture that would be stable for a few weeks. And passing it through guinea pigs again could restore its potency. Inoculating it into guinea pigs seemed to protect them from cholera, and he used it on rabbits and pigeons, too. That convinced him it could work in humans, and he inoculated himself. Barbara Hawgood writes:
The first vaccine did not produce any adverse reaction apart from light symptoms of fever (a small rise in body temperature and headache) and a local reaction of swelling and pain at the site of injection. These symptoms disappeared in a few days. Six days after the first injection, he was inoculated with the second cholera vaccine. Again the body temperature rose but returned to normal within 24 hours; local pain had disappeared by three days. With the safety of the vaccines demonstrated, Haffkine proceeded to vaccinate three Russian friends and other volunteers, one of whom was Mr EH Hankin, Fellow of St John’s College, Cambridge and recently appointed as chemical examiner, analyst and bacteriologist to the Northwest Provinces in India; again, only mild adverse effects were observed.
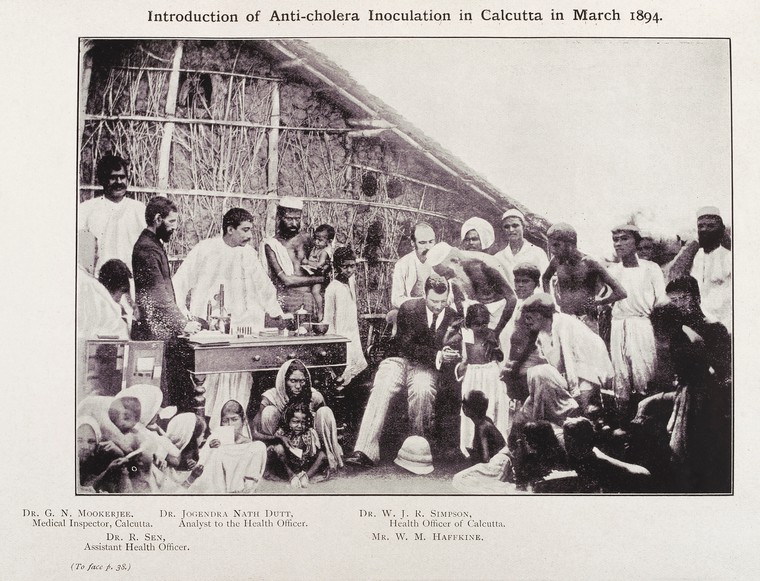
Haffkine went to India, where he vaccinated thousands of people in the military. But he wanted to do formal trial, with whole communities vaccinated and comparison ones not. Hawgood again:
Selection of the localities and groups of individuals was determined chiefly by the willingness of the people to undergo the preventive treatment; but, at the same time, in every place efforts were made to concentrate the operations on bodies of population living under similar conditions, and supervised by medical and sanitary authorities in order that, on the occasion of cholera outbreaks, a comparison could be made between the resistance of inoculated and uninoculated individuals.
But there was no cholera outbreak. Then he tried again, managing to persuade people to a smaller scale project during a cholera outbreak in Calcutta, including among prisoners. That attempt reduced illness, but not deaths.
Haffkine sickened with malaria and returned to Europe, but came back to India in 1896 and joined the civil service there. He developed a vaccine against bubonic plague, testing it in rabbits. His first-in-human test was himself again, this time with a higher dose than intended for others. The first test in others was again in prisoners. It worked well enough that 11,000 people were inoculated in 3 months in that outbreak.
Haffkine’s story also includes one of the times a vaccine went horribly wrong. To ramp up production in an outbreak where demand was high, he changed sterilization procedures, and a bottle of the vaccine was contaminated with tetanus.
A similar disaster happened with the polio vaccine in 1955, called the Cutter Incident, after the name of the company that prepared some vaccine tainted with live virus. (You can read about it in a Harper’s Magazine article from 1955.)
In 1963, the FDA introduced ground-breaking regulations for testing required to approve new drugs in the US. But as late as 2010, Karen Goetz and colleagues wrote, “Unfortunately, little if any specific guidance is available for first-in-human trials specific to vaccines”. It’s all hands on deck now for the Covid-19 trials, with agencies like the Health Products Regulatory Authority in Ireland issuing specific guidance this month. Laura Spinney reports that the Oslo-based non-profit Coalition for Epidemic Preparedness Innovations (CEPI) is financing and coordinating vaccine development. And the company that manufactured the vaccine in the Seattle trial has been working with the NIH on corona virus vaccine since the MERS epidemic.
If Covid-19 stays with us, a good vaccine could save a lot of lives. But it can’t arrive quickly, if it’s to arrive safely. At least these days, the people who sign up for the trials should be well-informed about what they are volunteering for. The rest of us will be very lucky that they were willing to do it. For most of the modern history of vaccine prevention, that may not have been case: neither the willing part, nor the well-informed part.
For our last tale, we go to the first time it’s known for sure that volunteers gave written informed consent to participating in research. That was in infectious disease, but it was about testing a hypothesis about transmission, not a vaccine. It was Walter Reed’s experiment that proved mosquitoes spread yellow fever, “The American Plague“.
It happened at the US Army’s Camp Lazear, that Reed named for a fellow physician, Jesse William Lazear. Lazear had died at the age of 34: he didn’t recover from the yellow fever he got after an infected mosquito bit him, presumed to be part of testing the hypothesis. The process involved breeding mosquitoes, letting them feed on people with yellow fever, and then letting them bite volunteers.
Army volunteers participated in these experiments, including ones that excluded the hypothesis that it was infected clothing that transmitted the disease. But they needed more volunteers, and so they offered money to locals: $100 to participate and an extra $100 to compensate if they contracted yellow fever. They received written contracts promising “the greatest care and the most skillful medical service from the Reed Commission”. When 4 people among a group of 12 volunteers contracted yellow fever in a week though, some decided the money was in no way worth it. Walter Reed wrote:
[S]everal of our good-natured Spanish friends…suddenly appeared to lose all interest in the progress of science, and forgetting for the moment even their own personal aggrandisement, incontinently severed their connection with Camp Lazear. Personally, while lamenting, to some extent, their departure, I could not but feel that in placing themselves beyond our control they are exercising the soundest of judgment!
Now, as then, it takes tremendous courage and altruism for people to provide health care and support to people suffering a dangerous infectious disease. In Spain, people in isolation are applauding their health care workers en masse, united through social media, to thank those who are protecting them from Covid-19. Being the first humans to test the vaccines is an act of courage and extraordinary generosity, too. Some day, many may have them to thank for their lives.
[Postscript 27 March 2020] Trials have now started on BCG vaccine, to see if it can improve people’s immunity to the coronavirus that causes Covid-19. The BCG vaccine is an anti-tuberculosis vaccine, that may have heterologous effects – TB may not be the only pathogens it can help people fight off. This is also called trained immunity. (More about that here.)
Jop de Vrieze reports that a randomized trial is starting this week in 1,000 healthcare workers in 8 Dutch hospitals, and includes background about the BCG vaccine in this context. You can read more about the trial in its trial registry entry. The primary endpoint for this trial is absenteeism, and the length is 180 days (about 6 months). Signs of infection with SARS-C0V-2 and symptoms of Covid-19 will also be measured.
A trial seeking 4,170 healthcare worker participants across several Australian hospitals was announced as well. It got off the ground so quickly, including ethics committee approval, because the Murdoch Children’s Research Institute in Melbourne is already running a trial of BCG vaccine to see if it can prevent allergies, and they were able to build from that. In May, the trialists got a grant for Aust$10m to expand it to 10,000 healthcare workers, including in Spain and the Netherlands. (You can see the trial registry entry for the healthcare worker trial here, and the one for the allergy trial here.)
[18 April 2020] Another 2 trials of BCG vaccine in healthcare workers reported to be starting soon: 700 healthcare workers in Boston and Texas; and 900 healthcare workers in Egypt.
[24 April 2020] A placebo-controlled trial of measles-mumps-rubella (MMR) vaccine to protect healthcare workers from Covid-19 is planned to begin in Cairo in May. It will aim to recruit 200 participants.
[25 April 2020] A trial of BCG vaccine in 1,000 healthcare workers in Medellín, Colombia was registered on 24 April.
[2 May 2020] Another trial of BCG vaccine, in 1,000 healthcare workers is planned to begin in June, in São Paulo, Brazil.
[5 May 2020] Another 2 trials of BCG vaccine in healthcare workers, one aiming to recruit 1,120 in France, aiming to recruit 1,500 healthcare workers in Denmark. BNT162 updated to include first dose given, and US trial register entry.
[8 May 2020] Another trial of BCG vaccine in healthcare workers was registered in South Africa, with 500 participants planned.
[15 May 2020] Another 2 trials of BCG in healthcare workers were registered, one in Hungary with 1,000 participants and one in Germany using VPM1002, developed from BCG vaccine, in 1,200 healthcare workers.
[8 June 2020] Late in May, another trial of BCG was registered: this time for 900 people 50 or older, in Greece. That brings the total number of people proposed for trials of BCG vaccines to protect against Covid-19 to 21,990 in up to 13 countries.
~~~~
 All my monthly Covid-19 Vaccine Race round-up posts
All my monthly Covid-19 Vaccine Race round-up posts
Disclosures: My only interest in Covid-19 trials is as a person worried about the virus, as one of my sons is immunocompromised. I have worked for an institute of the NIH in the past, but not NIAID. I am a contributor to the James Lind Library. More about me.
Note: At the time of writing this post, the trials registry, ClinicalTrials.gov, also had 2 entries for vaccines for prevention/treatment in people with Covid-19 in Shenzhen in China.
[Update shortly after posting] This post originally said I didn’t know what compensation Covid-19 trial participants in Seattle could expect.
[Updates 17 March 2020] Updated with NIH Director’s announcement that the first dose has been administered. The original post was: “with the first dose reportedly about to be injected shortly.” Also added the sentence on animal tests. Embedded link to Twitter of 2 participants in the Seattle trial talking about their involvement, and link to the interview with another.]
[Update 23 March 2020] Updated progress on Seattle trial – recruitment closed on 19 March.
[Update 24 March 2020] Updated addition of a site in Decatur, Georgia to the Seattle trial, and additional trial in China.
[Update 29 March 2020] Added Oxford vaccine trial. Thanks to Davina Ghersi for alerting me to its clinical trial registry entry. Based on that report, I changed “placebo vaccine” to “saline injection”.
[Update 31 March 2020] Updated progress on the Seattle/Decatur/Bethesda trial based on a 30 March change to its ClinicalTrials.gov entry.
[Update 11 April 2020] Added Phase 2 trial for Ad5-nCoV.
[Update 13 April 2020] Added preprint for pre-clinical work on INO-4800.
[Update 18 April 2020] Updated locations for the Oxford vaccine trial.
[Update 23 April 2020] Added Phase 1/2 trial for 4 BNT162 vaccines, and Phase 1/2 trial for Xuzhou trial. Added report of first participants injected in Oxford trial.
[Update 24 April 2020] Added Phase 1 oral vaccine trial in Canada, and MMR trial in Cairo.
[Update 25 April 2020] Added Colombian BCG trial.
[28 April 2020] Added Covid-19 S-Trimer trial.
[2 May 2020] Added Brisbane nanoparticle vaccine trial, BCG trial in São Paulo.
[5 May 2020] Added BCG trials in France and Denmark.
[8 May 2020] Update on mRNA-1273 going to phase 2 trial, and added South African BCG trial.
[15 May 2020] Added 2 more BCG trials: Hungary and Germany.
[17 May 2020] Added details of expansion of Australian BCG trial.
[8 June 2020] Added BCG trial in Greece.
The cartoons in this post are my own (CC BY-NC-ND license). (More cartoons at Statistically Funny and on Tumblr.)
Earnest Board’s painting of Edward Jenner vaccinating James Phipps via Wikimedia Commons.
The photo of the plaque at the house Edward Jenner bought for James Phipps was taken by Gaius Cornelius, via Wikimedia Commons.
The photo of Waldemar Haffkine and others in Calcutta in 1894 is via the Wellcome Collection.
The photo of Camp Lazear is from Howard Kelly’s 1906 book, Walter Reed and Yellow Fever, via the Internet Archive.




Very good article and helpful to understand the past trials of each n every vaccines
Thanks!
Superrrrb…Article ..
Amazing work…..
Thank you!
Gracias por subirlo!
I wish I could find a clinical trial looking for healthy individuals to volunteer for vaccine research relating to the current virus pandemic. None seem to be interested in a 63 year old.
Great article Hilda!
I am skeptical about the mRNA vaccine as I understand they require storage (and distribution) under -80c. This may be an extreme hurdle in getting doses in good condition to small clinics, not to mention rural areas and third world countries.
Hopefully they do find a way to make them stable under normally accepted temperatures.